Rintodestrant
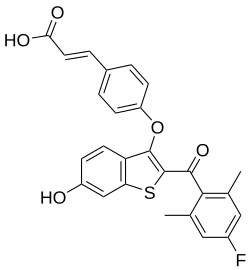 | |
| Clinical data | |
|---|---|
| Other names | G1T48 |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C26H19FO5S |
| Molar mass | 462.49 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Rintodestrant is an orally bioavailable selective estrogen receptor degrader (SERD) developed by G1 Therapeutics for the treatment of estrogen receptor-positive (ER+) breast cancer. Structurally inspired by the 6-OH-benzothiophene scaffold used in arzoxifene and raloxifene, rintodestrant selectively binds to the estrogen receptor and inhibits ER signaling, demonstrating efficacy in endocrine-resistant tumors.[1]
A phase I clinical trial evaluated rintodestrant as monotherapy and in combination with the CDK4/6 inhibitor palbociclib in patients with ER+/HER2- advanced breast cancer.[2]
References
- ^ Gheysen M, Punie K, Wildiers H, Neven P (November 2024). "Oral SERDs changing the scenery in hormone receptor positive breast cancer, a comprehensive review". Cancer Treatment Reviews. 130 102825. doi:10.1016/j.ctrv.2024.102825. PMID 39293125.
- ^ Clinical trial number NCT03455270 for "G1T48, an Oral SERD, Alone and in Combination With Palbociclib in ER-Positive, HER2-Negative Advanced Breast Cancer" at ClinicalTrials.gov