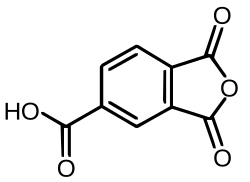
Trimellitic anhydride
 | |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.008.190 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H4O5 | |
| Molar mass | 192.126 g·mol−1 |
| Appearance | colorless or white solid |
| Density | 1.54 g/cm3 |
| Melting point | 168 °C (334 °F; 441 K) |
| Boiling point | 390 °C (734 °F; 663 K) |
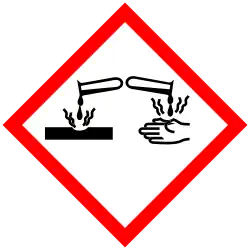
| Hazards | |
| GHS labelling: | |
| |
| Danger | |
| H317, H318, H334, H335 | |
| P261, P271, P272, P280, P285, P302+P352, P304+P340, P304+P341, P305+P351+P338, P310, P312, P321, P333+P313, P342+P311, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Trimellitic anhydride is an organic compound with the formula HO2CC6H3(C2O3). It is the cyclic anhydride of trimellitic acid. The compound is a colorless to white solid. It's primarily used in the production of various polymers, polyesters, agricultural chemicals and dyes. Several thousand tons of this compound are produced annually as a precursor to plasticizers for polyvinyl chloride. It is produced by air-oxidation of 1,2,4-trimethylbenzene.[1]
References
- ^ Röhrscheid, Freimund (2000). "Carboxylic Acids, Aromatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a05_249. ISBN 978-3-527-30673-2.