Cinolazepam
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Gerodorm 40mg |
| AHFS/Drugs.com | cinolazepam |
| Dependence liability | High |
| Addiction liability | Medium |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 90–100% |
| Metabolism | Hepatic |
| Onset of action | 30 minutes to 1 hour |
| Elimination half-life | 9 hours |
| Duration of action | 9 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
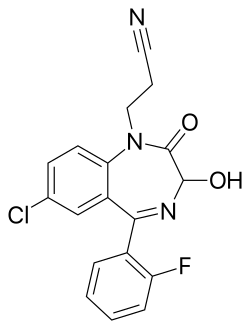
| Formula | C18H13ClFN3O2 |
| Molar mass | 357.77 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Cinolazepam[1] (marketed under the brand name Gerodorm)[2] is a drug which is a benzodiazepine derivative. It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties. Due to its strong sedative properties, it is primarily used as a hypnotic.
It was patented in 1978 and came into medical use in 1992.[3] Cinolazepam is mainly used in Romania and Slovakia; it not approved for sale in the United States or Canada.
References
- ^ US Patent 4388313 Novel 3-hydroxy-1,4-benzodiazepine-2-ones and process for the preparation thereof
- ^ Lannacher Romania (1999). "Gerodorm". Produse Gerot inregistrate in Romania (in Romanian). Archived from the original on 11 October 2006. Retrieved 17 August 2006.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 530. ISBN 9783527607495.