S33005
 | |
| Clinical data | |
|---|---|
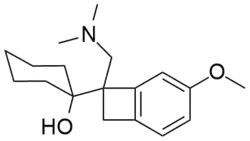
| Other names | (–)-1-(1-dimethylaminomethyl) 5-methoxybenzocyclobutan-1-yl) cyclohexanol |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C18H27NO2 |
| Molar mass | 289.419 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
S33005 is a serotonin–norepinephrine reuptake inhibitor (SNRI) that was under development by Servier for the treatment of depression and related disorders. It is structurally related to venlafaxine but has a more complex molecular structure. Venlafaxine appears to be a sigma modulator,[1] but it is not known if S33005 shares this activity.
Synthesis
"The 1-cyano-benzocyclobutenes used as starting material are obtained, for example, by subjecting a β-[orthohalogeno-phenyl]-propionitrile to intramolecular condensation in the presence of potassium amide, or by brominating a benzocyclobutene in position 1 with N-bromosuccinimide, followed by exchange of the bromine atom for a cyano group by means of sodium cyanide."[2]
See also
References
- ^ Dhir A, Kulkarni SK (June 2007). "Involvement of sigma-1 receptor modulation in the antidepressant action of venlafaxine". Neuroscience Letters. 420 (3): 204–8. doi:10.1016/j.neulet.2007.04.055. PMID 17532136. S2CID 25971336.
- ^ U.S. patent 3,622,614
External links
- PubMed search
- Binding Database
- The patent and synthesis discussion can be found in U.S. patent 6,107,345
| |||||||||||||||||||||
| |||||||||||||||||||||
| |||||||||||||||||||||
| |||||||||||||||||||||
| |||||||||||||||||||||
| Phenethylamines |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amphetamines |
| ||||||||||||||||
| Phentermines |
| ||||||||||||||||
| Cathinones |
| ||||||||||||||||
| Phenylisobutylamines (and further-extended) | |||||||||||||||||
| Catecholamines (and close relatives) |
| ||||||||||||||||
| Cyclized phenethylamines |
| ||||||||||||||||
| Related compounds |
| ||||||||||||||||
| |||||||||||||||||
This article is issued from Wikipedia. The text is available under Creative Commons Attribution-Share Alike 4.0 unless otherwise noted. Additional terms may apply for the media files.