3C-DFE
 | |
| Names | |
|---|---|
| Preferred IUPAC name
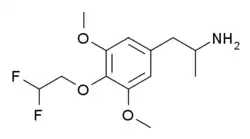
1-[4-(2,2-Difluoroethoxy)-3,5-dimethoxyphenyl]propan-2-amine | |
| Other names
3C-DFE
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C13H19FNO3 | |
| Molar mass | 275.292 g/mol |
| Melting point | 171–172 °C (340–342 °F; 444–445 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
3C-DFE is a lesser-known psychedelic drug, which is a fluorinated derivative of 3C-E. It was first synthesised by Daniel Trachsel in 2002,[1][2] and has been reported as showing similar psychedelic activity to related compounds, with a dose range of around 20–40 mg and a duration of approximately 10 hours.[3]: 736 Despite its reported psychedelic activity, binding studies in vitro showed 3C-DFE to have a surprisingly weak binding affinity of 2695 nM at 5-HT2A with negligible affinity at 5-HT2C,[3]: 737 making it only slightly higher affinity than mescaline, despite its higher potency in vivo.
See also
References
- ^ Trachsel, Daniel (2002). "Synthese von neuen (Phenylalkyl)aminen zur Untersuchung von Struktur-Aktivitätsbeziehungen, Mitteilung 1, Mescalin Derivate". Helvetica Chimica Acta. 85 (9): 3019–3026. doi:10.1002/1522-2675(200209)85:9<3019::AID-HLCA3019>3.0.CO;2-4.
- ^ Trachsel, Daniel (2012). "Fluorine in psychedelic phenethylamines". Drug Testing and Analysis. 4 (7–8): 577–90. doi:10.1002/dta.413. PMID 22374819.
- ^ a b Daniel Trachsel, David Lehmann and Christoph Enzensperger (2013). Phenethylamine Von der Struktur zur Funktion. Nachtschatten Verlag AG. ISBN 978-3-03788-700-4.
| Phenethylamines |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amphetamines |
| ||||||||||||||||
| Phentermines |
| ||||||||||||||||
| Cathinones |
| ||||||||||||||||
| Phenylisobutylamines (and further-extended) | |||||||||||||||||
| Catecholamines (and close relatives) |
| ||||||||||||||||
| Cyclized phenethylamines |
| ||||||||||||||||
| Related compounds |
| ||||||||||||||||
| |||||||||||||||||
This article is issued from Wikipedia. The text is available under Creative Commons Attribution-Share Alike 4.0 unless otherwise noted. Additional terms may apply for the media files.